

Nite Transportation Incubator
Unique Warming Technology
- The only transport incubator on the market to date with a servo-controlled radiant heating system
- Immediate and effective heat transfer to the newborn
- Best technology combined with a polyurethane resuscitation bag
- Temperature regulation in air or skin mode
Facilitates the work of caregivers
- Intended for the transport of newborns between departments within the hospital (internal transfer) and between different hospital establishments (land or air transport)
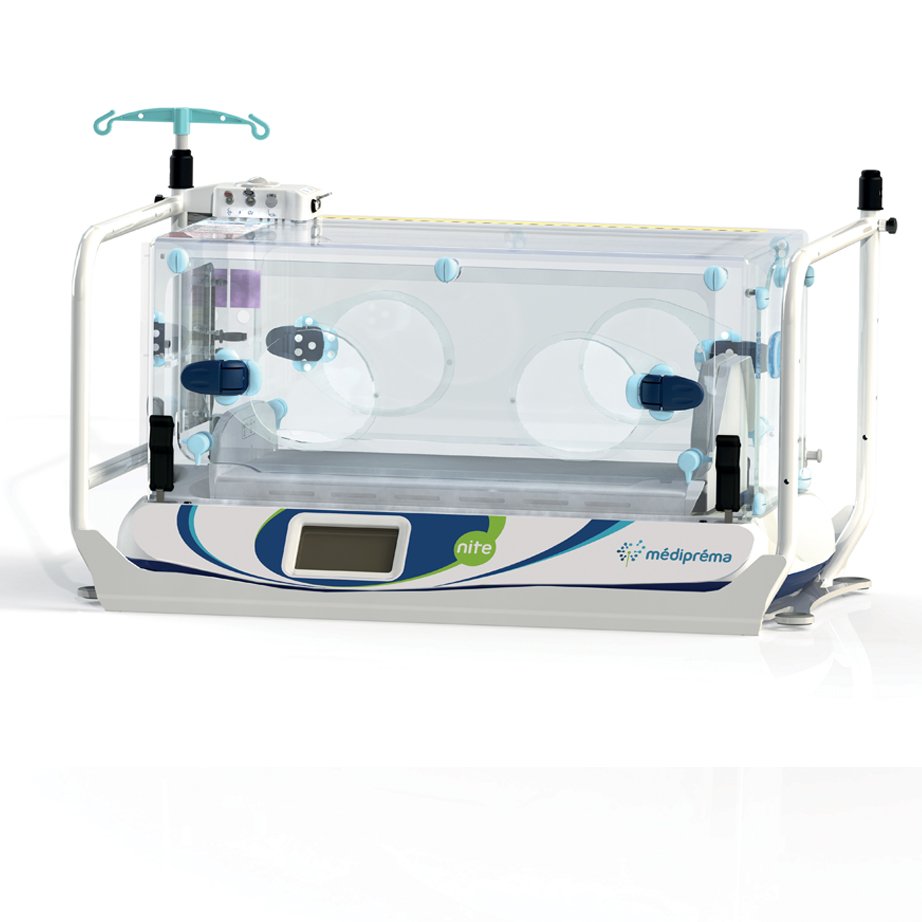
- Ergonomic: perfect visibility of the child on all 5 sides, total accessibility to the child from all 4 sides, lightweight (37kg)
- User-friendly touchscreen
- Automatic power source recognition
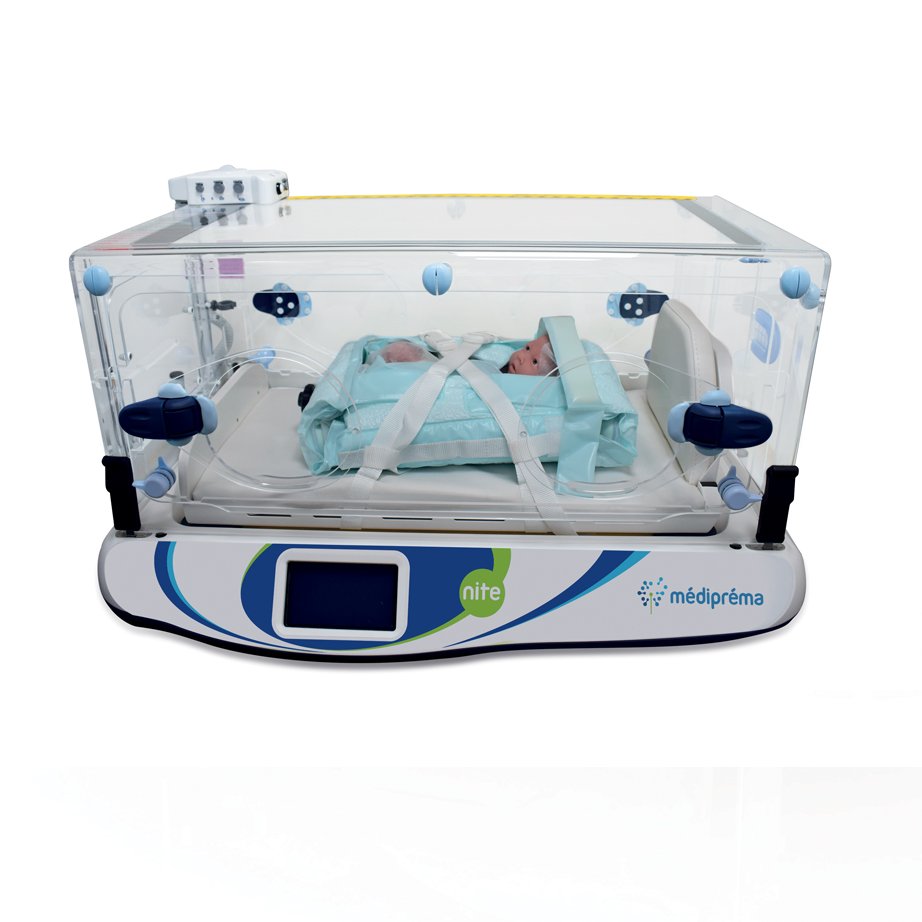
Child comfort
- Rapid warm-up
- Isolation from the external environment
- Maintaining the control temperature in all circumstances (e.g. possibility of fitting and resuscitating the child with the doors open while warming him/her)
- Silicone feet of the sleeping surface to limit unpleasant vibrations for the child
- Spacious interior
Unique warming technology
The Nite transport structure by médipréma has a real radiant vault heater, integrated into the incubator cabin, ensuring immediate heat transfer to the newborn. The thermal insulation has been designed to protect the child from environmental disturbances and numerous door openings during transport.
This heating system, effective in all circumstances, makes it possible to intervene on the newborn with the doors open without compromising its warming. The temperature rise is very fast (20min from 25°C to 36°C, control temperature 37°C), thus guaranteeing a rapid warming of the newborn. The presence of an air temperature measurement sensor in the passenger compartment combined with the vault temperature measurement probes makes it possible to perfectly modulate the heating, which operates safely regardless of environmental conditions.
Ergonomics designed to facilitate the work of medical teams
Like the other medipréma incubators, the interior of the Nite transport structure has been designed to guarantee total accessibility to the child. Equipped with 2 large doors with portholes on the front and back, a hatch on the foot side and a double door on the head side, it offers total accessibility to the child from the 4 sides of the incubator. It also allows perfect visibility of the child on 5 sides thanks to its transparent plexiglass interior.
The Nite transport incubator by médipréma is equipped with a touchscreen and intuitive LCD display (with the possibility of selecting different working languages) that allows a good visualization, with zoom, of the temperature curves in real time (monitoring) and their history.
A reliable solution without compromising child comfort
The Nite incubator by médipréma has been designed to improve the conditions of transport of newborns. The incubator’s sleeping surface is placed on silicone feet to limit vibrations related to transport. The sleeping surface is reversible (head, feet) to adapt the position of the child to the equipment on board and the position of the caregiver. The interior has been designed to limit noise pollution from the fan as much as possible for more peace of mind for the newborn.
Many accessories are available to improve the comfort of the child and the medical teams during transport: harness to hold the child, vacuum mattress, proclive cushion, protective bar, etc.
PERFORMANCE
Temperature regulation
- air from 28 to 39°C (with safe extension beyond 37°C)
- 35 to 38°C (with safe extension beyond 37°C)
Main heating by radiant vault integrated into the passenger compartment with heat recovery located under the sleeping surface
Presence of an air temperature measurement sensor in the passenger compartment which, combined with the vault temperature measurement probes, allows the heating to be perfectly modulated, which works safely regardless of the environmental conditions
Heating power: 330 watts (power consumption: 450 VA)
Built-in heating battery (15 to 45 minutes of autonomy): Possibility of adding an external battery to increase this autonomy up to 4 hours
Multi-source power supply: alternating current 110 to 240V, 50/60/400Hz and continuous current from 12 to 36 V
Electrical class: II
DESCRIPTION
Display of instructions on a touchscreen LCD screen with the possibility of selecting the working language (French, English, German or Spanish). Zoomed visualization of real-time temperature curves (monitoring) and their history
Very spacious plexiglass interior with perfect visibility on 5 sides
Total accessibility to the newborn from all 4 sides:
- Front: 1 large door with 2 portholes
- Rear side: 1 large door with 2 portholes
- Foot side: 1 hatch
- Head side: 1 double door
Optional 70% outgoing tray, for full access to the child’s body (easily interchangeable with the standard tray)
Fold-down headrest for easy access to the child’s head (standard tray only)
Sleeping surface: 610x340mm
- Placed on silicone feet to limit vibrations, held in the incubator box by unlockable fixing indexes
- Reversible sleeping surface (head, feet) to adapt the position of the child to the on-board equipment and the position of the caregiver
Socket for surveillance lighting
4 built-in handles
Overall Dimensions (LxWxH): 910x580x518mm
Weight: 37kg
4 attachment points on the structure
OPTIONS & ACCESSORIES
- Probe for integrated FiO2 analyzer
- Protective and transport bar with serum holder rod
- Outgoing tray
- Insulated cover
- Foam/microbead mattress for transport
- External battery pack
- Respirator hose support arm
- 10G Newborn Support Solutions
- Integration on Médipréma road, helicopter or intra-hospital transport modules
Class IIb
Medical Device Electrical Class II